
Within the coming weeks, we should be starting to get a picture of whether remdesivir will be a savior to the world, just another failed drug, or something in between.
There are already clues in a March 24th paper in PNAS called "Prophylactic and therapeutic remdesivir (GS-5734) treatment in the rhesus macaque model of MERS-CoV infection."
Something should stand out right away. The paper isn't studying the "right" virus; the title says MERS rather than SARS – the bug that is crippling us now. Perhaps worse still, remdesivir was discovered as a treatment for Ebola but failed. So isn't it vaguely disturbing that Gilead appears to be simply grasping at straws, hoping that it might work for something? No, it isn't.
1. Yes, it makes sense to study MERS
At the onset of the epidemic, remdesivir was already being studied in mouse and in vitro models of MERS, another coronavirus that was first identified in 2012.
But in order to have a chance of stopping SARS-CoV-2, the needlessly complex name given to what we just call "coronavirus", remdesivir needs to at the very least be able to stop the replication of the virus in a "test tube" as it did with MERS.
It does (Figure 1). Quite well.
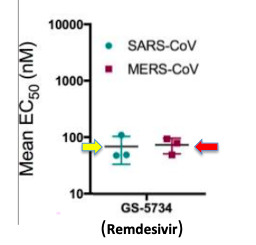
(Figure 1) The EC50 values (lower means more potent) of SARS (yellow arrow) and MERS (red arrow) are both approximately 100 nM (nanomolar), which is equivalent to 0.1 micromolar. 100 nM inhibitors are considered to be potent.
Remdesivir's effect on pre-treated and therapeutically treated animals
Animal models of human diseases vary widely in their ability to predict whether a drug will work in people. In general, bacterial and viral models are fairly predictive of efficacy in humans, and when the experiment is run in monkeys rather than rats, the relevance to human disease is greater still.
Procedure (Figure 2)
Eighteen rhesus macaques were divided into three groups, each containing 6 monkeys.
Group 1 - Three monkeys received only the vehicle solution (without drug) 24 hours prior to being infected and three more received the placebo 12 hours prior to infection.
Group 2 - Six monkeys received 5 mg of remdesivir per kilo of body weight 24 hours before being infected.
Group 3 - Six monkeys were treated with 5 mg of remdesivir per kilo of body weight 12 hours after being infected. Treatment was continued for six days and all 18 animals were euthanized.
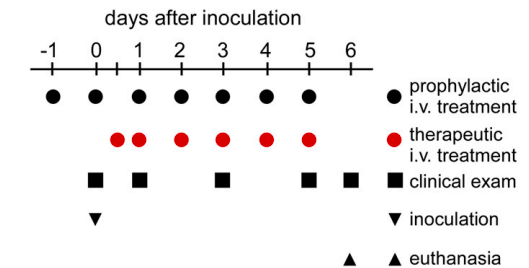
Figure 2. Experimental timeline. (Black circles, group 2) Prophylactic treatment began one day prior to inoculation. (Red circles, group 3) Therapeutic treatment was begun 12 hours post-infection, 12 hours later, and then once per day. The black squares indicate clinical exams (every day).
Results (prophylactic treatment)
When the monkeys were treated with remdesivir 24 hours before they were inoculated (infected) the drug completely protected them from clinical signs of MERS disease. The pre-treated monkeys were also protected from MERS lung lesions, and the amount of viral replication in the lungs – the hallmark of the infection – was strongly inhibited.
Results (therapeutic treatment)
Although prophylactic use (black) of the drug resulted in almost complete protection of the animals, the data on therapeutic use (red) are less dramatic. It is clear that the drug works better prophylactically than therapeutically. Remdesivir reduced the viral load in respiratory tissues (left) and improved lung health as measured by x-ray. The control group is shown in gray (Figure 3). 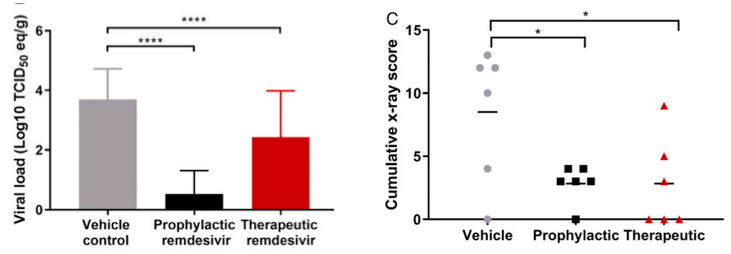
The same trend was seen in other mucosal tissues (Figure 4).
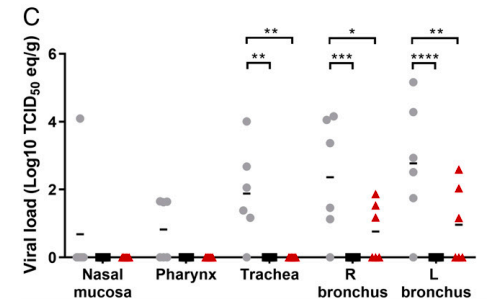
Figure 4. Viral load in different mucosal tissues.
Source for all figures: PNAS
Summary
Remdesivir passed the monkey tests with (more or less) flying colors. Even though the drug was tested against MERS, not SARS, it inhibits both viruses with similar potency. So MERS should be a good surrogate for the current coronavirus. What isn't clear is what the drug will do once a person is already infected. It won't clear all the virus from the lungs but it may slow down the replication so that the disease is less serious. Only human trials can determine this.
It is clear that, as the case with other antiviral drugs such as Tamiflu and Valtrex, the earlier remdesivir is given the better it works. This is consistent with the mechanism of the drug – inhibition of viral RNA synthesis, which is an early process in RNA viral replication.
If the inhibition of coronovirus in humans by remdesivir is consistent with that of MERS in rhesus macaques, we will have our first evidence-based tool against the virus. And not a moment too soon.
Disclaimer: I have a (pitifully) small amount of Gilead Stock in my IRA.



